There are several important differences between hydrogen bonds (H-bonds) and classical chemical bonds. Some excellent information is on Chaplin’s site: https://water.lsbu.ac.uk/water/water_hydrogen_bonding.html
Models for describing gas phase intermolecular interactions include the classical van der Waals (VDW) interaction among many others. As well, there are different models for condensed phase interactions. One frequently used model is the Lennard-Jones (6-12) potential, in which repulsion has a 12th power dependence on separation, and attraction a 6th. Described in this post is the coulombic distance dependence of H-bonds, which can be viewed as largely electrostatic. Generally, while weaker than conventional bonds, H-bonds extend over far greater distances.
If we have a water molecule with fixed charges of approximately +0.32 on H and -0.64 on O we can model the distance dependence of the H-bond energy of a pair of waters. Each atom in each molecule of the dimer has a charge and a location. We label the charges in molecule 1 as j, for j=1,3, and label the atoms on the molecule 2 with k, k=1,3 and the distance between atoms on different molecules is Rj,k.
Total electrostatic energy, E/(coulomb constant C), is then:

Starting with a water dimer, we now separate the molecules along a vector defined by the two oxygen molecules maintaining the geometry of the hydrogens relative to the oxygens and the orientation relative to the vector. This procedure is not accurate, but it is easy to understand. While the H-bond is weaker than a classical bond, it extends over far greater distances. The much greater “reach” of the H-bond is how vibrations between molecules are coupled between multiple pairs of atoms. The average separation between water molecules is just less than 3 Angstroms. The value of KT at 300oK is around 2.5kJ/mole. Included for comparison are Mopac22(PM6-ORG) results where the only geometric constraint was the separation between the two oxygen molecules. Historically many molecular models did not include electrostatic interactions other than bimolecular.
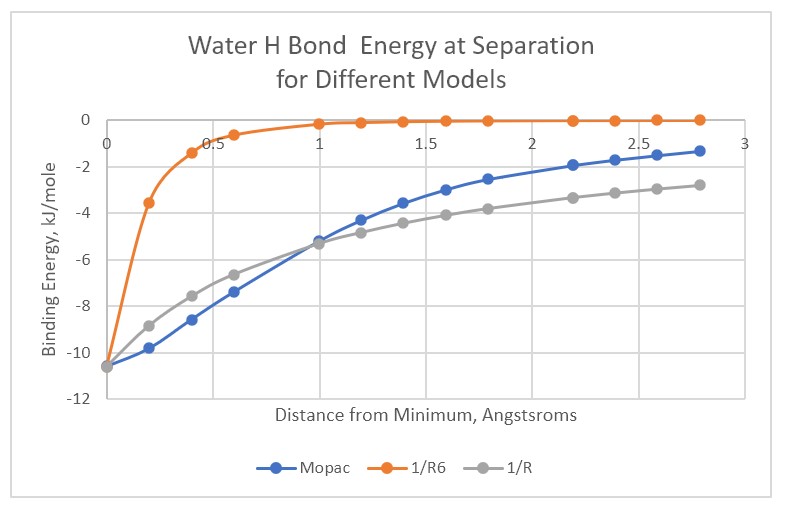
The long-distance interaction means that water molecules can interact over distances greater than the average separation of water molecules. The effective volume of interaction of a water molecule must be much larger than that attributable to the (6-12) volume. The greater volume also implies more competing entropic states. As there are many possible entropic states to compete with any particular low energy state, H-bond geometry is not well defined. Different experiments give different values for the bond energy. Given the variety of entropic states, it is similarly difficult to define temperature for any particular experiment when one process is seeing Maxwellian statistics while another is seeing Bose-Einstein as is the case for vibration energies.
Energy Minimum Coordinates and Charges for H2O2 using Mopac22 PM6-ORG
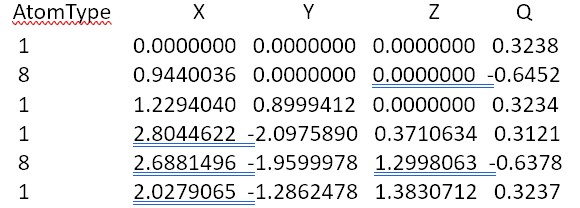
The O-O distance is 2.92 Å
View this article as a PDF file.
Critical comments accepted.